The LNP Mystery Part II
An examination of Dr. Palmer's brief assessment.
This is a continuation, albeit somewhat topically separate post from PART I
Note: An update has been added to part of the information below.
Scouring the internet and clicking on whatever link I can find about LNPs, I came across this video from Dr. Michael Palmer. I’ve never heard of him, but I’m always interested in seeing what other perspectives are out there. For those who do not know, Dr. Palmer is a medically trained doctor who works in pharmacology research and is positioned at the University of Waterloo.
Now, if it isn’t quite clear yet I have not been entirely convinced that these LNPs are anything more than a red herring- a possible player in the sea of vaccine adverse events that may not actually have a starring role. For now, it appears these LNPs may be more along for the ride rather than the agents of chaos we are searching for.
Dr. Palmer thinks otherwise, so it was nice to see what his assessments are of these LNPs. His video is short and brings up a few points worth considering. I must also comment that it’s several months old, and so his thinking may have changed since the time of this recording and airing.
The cationic nature of the lipids are toxic.
Will disrupt mitochondrial function and produce reactive oxygen species which may be damaging to DNA.
Okay, so it’s a rather short video that didn’t go too far in depth, but I would rather take this over sifting through hours of Dr. Ardis’ videos! What’s more important is that Dr. Palmer apparently provides a few citations for his argument.
Therefore, it will be worth looking at some of these papers and see if we can really find any evidence of concerning toxicity.
What are Lipid Nanoparticles?
So the acronym LNP has been thrown around a lot. Some of you still may not know what it stands for, but in short it refers to Lipid NanoParticles, which are structures that encapsulate the mRNA of the Pfizer and Moderna vaccines.

LNPs are considered the most recent form of vaccine development, gaining more notoriety compared to viral-vector vaccines1.
What’s important about LNPs is that the LNPs of these vaccines refer to the complex structure formed by various lipid molecules.
For instance, Pfizer’s COVID vaccine contains the following lipids in their LNP formulation: 0.43mg ALC-0315, 0.05mg Polyethylene Glycol (PEG), 0.09mg DSPC, and 0.2mg Cholesterol. The two primary lipids in question have been bolded.
Moderna’s COVID vaccine contains all of the same lipids as Pfizer’s except for the ALC-0315 which was replaced with the controversial SM-1022.
Even though the idea of a lipid structure seems simple on the surface, it is actually quite complex. The lipids need to keep their integrity so that the mRNA does not become released too early and also degrade. Therefore, the ratio of lipids as well as the chemical structures of individual lipids are important. The addition of both PEG and Cholesterol are intended to aid in that structural integrity.
What’s rather unique about the LNP formulations for these vaccines is their interaction with the mRNA. Normally, the cationic nature of these lipids was considered adequate enough to interact to the negatively charged phosphate backbone of the mRNA. However, recent formulations, such as those seen in ALC-0315 and SM-102 have an additional little quirk.
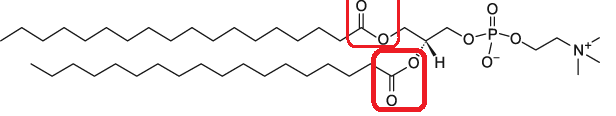
In the case of SM-102, one of the carbon chains attacked to the central Nitrogen is an Alcohol group. This group, circled in blue is called an ethyl hydroxylamine group (butyl hydroxylamine group for ALC-0315 since it is a 4-Carbon chain).
This little addition, although minor, dangles into the center of the liposomes and is able to engage in hydrogen bonding with the mRNA to provide for more stability of the structure. It also compacts the mRNA, which supposedly increases the transfection rate of these LNPs. Overall, the structure of the LNPs used within these vaccines are relatively new, and although their modifications are intended to be improvements upon prior LNP formulations there is no robust literature describing these LNPs.
Reviewing the Studies
It may come as no surprised, but the published literature on lipids is rather scant, so it’s hard to really discern much of the information out there.
Nonetheless, it appears that Dr. Palmer based most of his hypotheses predominately on 4 papers.
The two most important papers are the two review papers provided.
Toxicity of Cationic Lipid-DNA Complexes (Yew, N. S. & Scheule, R. K.)
The first review article is one from Yew, N. S. & Scheule, R. K., and as the title suggests this article looks at LNP-DNA complexes, and so we may not be able to draw similar conclusions from these studies to the mRNA vaccines.
This review also doesn’t look at intramuscular injections, and instead looked at intrapulmonary and intravenous injections. Now, there are concerns about the LNPs entering into the bloodstream, and from that perspective we may be able to draw similar concerns over LNP distribution. We already have evidence from the Pfizer trials about LNPs travelling (which I have written about before). In some sense, we may be able to extrapolate a bit using consilience, but again care must be taken to not draw too far from the information provided.
For example, the review does indicate that LNPs may interact with serum proteins due to their cationic nature and disrupt cells in the bloodstream:
Cationic lipid-DNA complexes interact with serum proteins, erythrocytes, monocytes, and other blood components upon IV injection. Some of the complex-binding proteins that have been identified include apolipoproteins, fibrinogen, and several others (Tandia et al., 2003). Complexes aggregate immediately upon exposure to serum, and also interact with red blood cells, causing extensive agglutination (Eliyahu et al., 2002). Prolonged interaction with serum causes disintegration of the complex (Li et al., 1999a). In addition, cationic lipids such as DOTMA/DOPE can induce fusion between erythrocytes (Sakurai et al., 2002).
The reviewers also noted that the liver is one of the primary collection points of the LNPs, as noted within the Pfizer documents.
The issue here, is to what extent these actions are occurring with the LNPs used in these vaccines, as the formulation used for the COVID vaccines does not reflect the ones listed within this review which appears to have only examined liposomes made from one lipid. We should keep in mind that this review article is from 2005, at a time in which most non-viral vector therapeutics were developed for either cancer treatment or drug delivery.
As we will find out, the structure of the LNPs is where a lot of the toxicity is derived.
Overall, this review does raise questions in regards to the actual damage that may arise from circulation of these LNPs. However, it only provides a possible speculative direction.
Toxicity of cationic lipids and cationic polymers in gene delivery (Lv, et. al.)
The second review article is a lot more appropriate to our situation, as it actually outlines the concerning features of the lipids used in these LNPs.
For instance, the reviewers outlined a few critical points when examining the structure of these lipids:
The cationic nature of the head groups
The makeup of the tails- we can skip this, as most formulations of lipids use relatively similar tails. Also, as the reviewers noted, the toxicity of these lipids are likely to be based on the other two groups.
The linker groups
A schematic representation of these lipid features can be seen below:
Head Groups
Starting off, most research into lipid toxicity has found that the hydrophilic head group greatly affects toxicity. Dr. Malone has indicated this before, and discussed the toxicity of quaternary amines in lipid formulation.
Previously, I mentioned that the head group of SM-102, as well as ALC-0315 is an ionizable cationic head. This means that the Nitrogen head group has 3 bonds and 1 lone pair, which allows it to bind to free-roaming hydrogen3. In doing so, the head will become cationic in nature. However, this interchange means that the cationic nature can be added or removed, thus making the head group ionizable and subject to change based on the environment.
With quaternary amines, the Nitrogen actually has 4 bonds to Carbon chains. In doing so, these head groups are not ionizable, because the cationic nature cannot be removed and the positive charge is permanent. This is likely why quaternary amines are so toxic.
Linker Groups
Linker groups connect the Nitrogen-based head group to the hydrophobic tail. These groups are important because they indicate how biodegradable4 the lipid is, and whether it is subject to easy breakdown or may persist and lead to toxic damage.
As the reviewers noted, ester groups (circled in red in the above molecules) and carbamate linker groups (not shown) are the best at decomposing due to their reduced stability compared to amides and ether groups.
What this means for the vaccine lipids
Taken together on just the individual structures alone, the lipids used in this vaccines should be relatively- RELATIVELY, less toxic than those used in prior formulations. The head groups of both SM-102 and ALC-0315 are ionizable, tertiary amines and thus should be less toxic than their quaternary counterparts. Although DSPC is a quaternary amine, its content within the LNP is far less than that of the tertiary amines.
All 3 lipids also contain ester groups for their linker groups, which would also suggest a higher degree of biodegradability and thus less overall toxicity.
In comparison, most of the lipids detailed in the Yew, N. S. & Scheule, R. K. review were quaternary amines with ether linkages such as DOTMA, and so in that context it may explain the high level of toxicity outlined in that review.
So does this mean that the COVID vaccine lipids are out of the woods in terms of toxicity? Not necessarily. Even though the individual lipids may be relatively safer than prior lipid designs, the overall LNP complex cannot be overlooked. The dynamics between the lipids as well as the mRNA may change the behavior of these complexes as well as their interactions within our bodies, which are all important factors to consider when examining the overall toxicity of these products.
It’s the entirety of the structure, as well as the individual lipids themselves that are of concern. Unfortunately, most studies either do not make that difference or examine these nuances extensively.
In Vitro Studies
This leaves us with the last two studies in question, which are the studies by Soenen, et. al.5 and Cui, et. al.6
I won’t address these studies in full, but these studies incubated cells with different liposome formulations and examined the effects on cell viability and the production of Reactive Oxygen Species (ROS).
Now, I won’t criticize these studies for being in vitro as the study must be examined within that context. However, there are few issues with these results and how they relate to the vaccines we are seeing:
Lipid Structure: We’ve already discussed the importance of the lipid structure in regards to their toxicity, and the researchers of both studies have indicated such as well (the latter study focused specifically on the toxicity of the headgroups). The issue here is that neither studies used tertiary amine lipids in their studies- the ones found in the COVID vaccines, so we can’t quite relate the results of these studies to what we are seeing now.
Liposome Makeup: Both studies also constructed liposomes that were comprised of only one or two lipids (usually quaternary amines), and once again are not reflective of the LNPs we are seeing. Why are the formulations important? The Lv, et. al. study suggested that PEG may help to stabilize the liposome from degradation and may actually contribute to reduced toxicity. So we may not be able to extrapolate out to relate to concerns over COVID vaccines.
The “so what” phenomenon: Both studies looked at the production of ROS, and may be related to endosomal uptake of the lipids and possible disruption of mitochondrial function. On the surface that’s rather concerning, and both studies suggest that this may be a possibility with these synthetic lipids (remember more specifically with the quaternary amines). Production of ROS may be concerning because it may lead to eventual cellular apoptosis and therefore extensive cell death may be concerning. However, when we look at it within the context of these vaccines, we have to keep in mind that these LNPs are essentially the “kiss of death” for cells- any cells that take up these LNPs may eventually produce spike proteins, present them on their cell surface, and then be targeted for death. So on that note, is it really concerning that ROS may be produced if these vaccines would lead to cell death anyways? Essentially, the production of ROS may be considered a “nonissue” since that would likely happen regardless if the causative agent was the lipid or the spike protein presentation.
Note: After giving this further thought I believe I have jumped the shark on this assessment. I will release an update either today or tomorrow outlining the issues with this thinking, but I will leave this bullet up for posterity and transparency.
The last note is especially interesting. Dr. Palmer describes the production of ROS as being a concern, but that is likely based upon the two studies above which may not relate to the current situation. What’s rather strange is how Dr. Palmer alludes to the production of ROS and the downstream damage as being similar to that of gamma or X-Ray radiation. That sounds extremely concerning, but considering that many people experience ROS caused by many different disease states, including obesity and diabetes, I wonder why the choice of gamma or X-Ray radiation was chosen, if not for the possibility of stoking some from unnecessary histrionics (take a look at one of the YouTube comments on the ROS remark and see how people replied to get an example).
What can we gather on LNPs
Even something as simple as a carrier agent comes with its own complexities. Right now, as we struggle to elucidate where all of these adverse reactions are coming from, we may point fingers at anything without necessarily understanding if our concerns are warranted.
I have held the belief that the LNPs may be a red herring for blaming the carrier, rather than the package, but an evaluation of some of these studies does suggest possible toxicities, of which we hardly have any data on as of this moment.
In that regard, caution is needed to extrapolate too far based on the studies presented, although we must still be open to further assessment and analyses of the information.
For now, I find that these studies are interesting, and they likely allude to possible mechanisms that should be examined further. But they also don’t detail exactly what is happening with these vaccines. The lipids are not correct, the LNP/mRNA complex has not been extensively examined, and we need all of these in order to gain a proper insight. Even then, the lipids used in these vaccines may be considered relatively safer, but within the broader scope how safe are cationic lipids overall?
Keep in mind that this assessment was narrowed to the scope of the studies provided in the video of Dr. Palmer, and there are a few studies out there that may prove that these LNPs may be far more dangerous than is being presented. I’m open to seeing other studies, and I encourage others to post studies that may be worth examining.
I’ll open the floor to people who have additional concerns or questions, and I’ll review more studies as they begin to come in.
Viral-vector vaccines suffer from an issue of them virtually being viral vectors: if a person has prior immunity to the viral vector it is likely that the vector would be targeted and eliminated before administering the antigen-containing genetic material. LNPs as a vector of delivery helps to overcome that prior immunity burden.
Unfortunately, I could not find the actual amount of the individual lipids. The FDA product insert only suggests that the total lipid count in a 0.5 mL dose is 1.93 mg total. However, we may have to assume that the structure of the LNPs have some optimized proportion, and thus may be similar to that of Pfizer’s.
Remember that this function is due to the pKa of amines. Typical amines have a pKa around 9 while physiological pH is close to 7. Because the pKa of amines are higher than physiological pH, they tend to be protonated and thus cationic in nature. Strangely, the pKa for these lipids may actually be closer to 7 rather than 9.
This isn’t the same “biodegradable” that is used when discussing environmentally-friendly products, although the root of the term may be similar. Unlike decomposition within the environment, biodegradable in this sense refers to the breakdown- either spontaneously or through enzymatic reactions- of the lipids. Therefore, it refers to cellular activity that may cleave and cut up the lipids in question.
Soenen, S. J., Brisson, A. R., & De Cuyper, M. (2009). Addressing the problem of cationic lipid-mediated toxicity: the magnetoliposome model. Biomaterials, 30(22), 3691–3701. https://doi.org/10.1016/j.biomaterials.2009.03.040
Cui, S., Wang, Y., Gong, Y., Lin, X., Zhao, Y., Zhi, D., Zhou, Q., & Zhang, S. (2018). Correlation of the cytotoxic effects of cationic lipids with their headgroups. Toxicology research, 7(3), 473–479. https://doi.org/10.1039/c8tx00005k





So this is good for understanding LNPs as we are basically under the impression that they are just fat droplets which sounds relatively harmless. In reading this I thought about the Quat disinfectant that we have in the lab which is a Quaternary amine (which I now know has a permanent positive charge which would allow it to bond/penetrate a negatively charged microorganism).
I found a couple more Dr. Palmer videos on Odysee which I am in the process of watching. It's all very interesting and I really think the main thing we are learning is that we have no idea what is going on in the body when these things are injected. We have so many poorly designed/carried out studies (TOGETHER trial or pretty much all the released Pfizer studies) which are being touted as absolute evidence. There are SO many things to consider when injecting these things into our bodies and it seems almost none of them have been considered (or if they were they were just glossed over).
ROS is also quite interesting and I have been hearing a lot about it lately so I am looking forward to your upcoming re-assessment.
Ok, you're still not believing it's the lipids alone?
Here's a good tidbit:
Moderna, pre COVID , during their gene therapies research had to stop because they were having issues with subsequent doses. So they shifted to vaccines (thinking single dose or 2 doses).
They used the same lnp, licensed from arcuitis (and pfizer uses it too). These lipids are the common factor, as the past issues happened before any research on it to deal with viruses.
But moderna claims that they made their own lipids. But they declared this after their trial was approved to go , which was based on the licensed lnp.
Whitney Webb exposed this moderna stuff, and she thinks moderna is lying, and the licensee is not suing yet because they didn't want to be looked on badly during the "pandemic". I think she's spot on.