SARS-COV2 is not a neuroinvasive virus?
Mounting evidence may refute the prevailing notion that SARS-COV2 infections lead to direct neuronal damage.
The following article does not refute the idea that SARS-COV2 does not directly invade the nervous system. Instead, it argues that this event likely is not occurring at the frequency many may assume, and in fact many signs of SARS-COV2 may be associated with other cellular mechanism rather than direct neuronal damage. Remember that several possibilities are possible, and many are likely to overlap.
Edit: A citation was missing for the microglial excerpt. It has been added along with a link to the article.
Does SARS-COV2 target the nervous system?
Surprisingly, it appears that one of the longest held-beliefs in regards to SARS-COV2 may not actually be substantiated.
That is to say, it doesn’t appear that SARS-COV2 actually directly targets the nervous system in many cases.
Most of this evidence comes from various pieces of literature from autopsy reports that don’t actually note direct damage to neurons, and instead notes damage nearby regions.
For instance, the study from Nuovo, et al.1 in which the brains of 5 dementia patients (4 Alzheimer’s and 1 with Lewy body dementia) as well as the brains of undemented COVID-19 patients were assessed for CNS damage.
Instead of finding evidence of SARS-COV2 or other viral proteins, the researchers found that most histology samples noted spike proteins but either no or very rare instances other proteins such as nucleocapsid proteins or viral RNA in the brains of those with COVID (in those with and without dementia).
More importantly, the spike proteins were found around the endothelium (blood vessels) within the brain rather than the brain itself, with the uptake of spike by endothelial cells leading to endothelial dysfunction and neuroinflammation from microglia.
Again, strikingly, the movement of exogenous immune cells did not appear to occur, but rather the response to spike was dictated predominately by microglia in the brain as proposed by the following schematic:
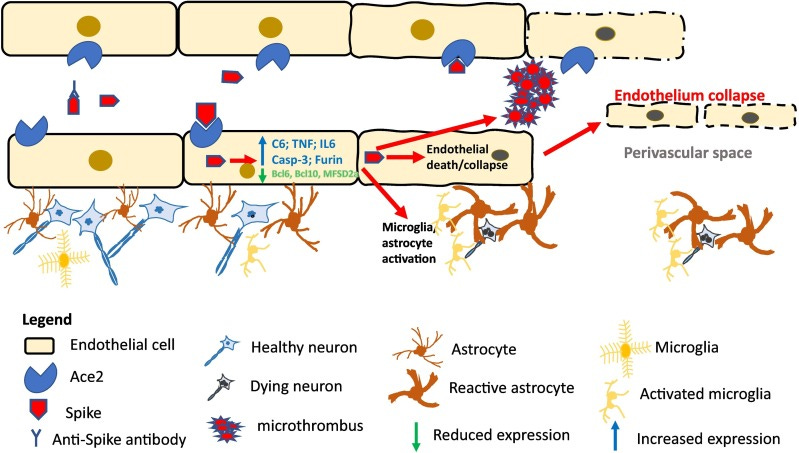
What about spike in the brain?
This study from Nuovo, et al. alone cannot refute the idea that SARS-COV2 does not target the brain directly, or that spike protein may find its way into the brain and cause direct cytotoxic damage.
There’s the neuropathology report2 provided by Dr. Oliver which looked at brains of those who died of COVID and noted spike and nucleocapsid protein the brains of some individuals, as well as some of the nerves, as can be seen in the following slide:
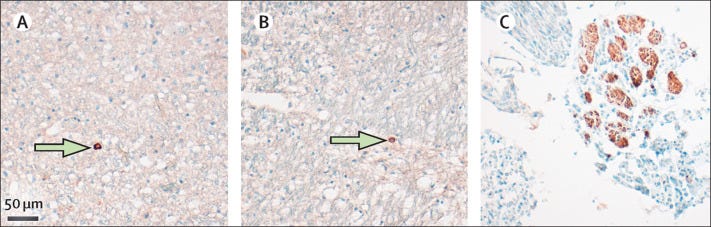
It’s important to remember that nearly all of the patients had pneumonia recorded as a cause of death, and nearly all patients had comorbidities such as heart disease, Parkinson’s disease, hypertension, and diabetes.
Essentially, these aren’t the healthiest of patients, and so it’s possible that their comorbidities may have contributed to endothelial dysfunction and blood-brain barrier permeability.
In that case, the detection of viral proteins serve as a red herring in some sense, only being found after severe infection and systemic damage has occurred that allowed movement of these proteins and viral RNA.
Put another way, the presence of spike or nucleocapsid protein may not be indicative of neuropathological changes in the brain- the body may have suffered enough damage that allowed the passage of these proteins.
This is part of a larger issue of science in which a bystander protein or molecule is used as an indication of cell damage and death. As was covered previously, the presence of tau proteins, amyloid plaques, and Lewy bodies does not indicate that these proteins are toxic themselves, and in fact may be produced in response to some pathogen and done in an attempt to protect the brain.
More recently, a widely spread article on a sugar-substitute erythritol made comments that elevated erythritol levels may be associated with increased risk of heart attacks and strokes when this compound may, in fact, be produced by cells in higher numbers as a consequence of cardiometabolic disease.
But what about anosmia and the olfactory bulb?
Anosmia, or the loss of smell, has been used to great extent to suggest that neurons within the olfactory bulb may be targeted by SARS-COV2.
This would be in line with the Braak hypothesis and a nasal route of entry into the nervous system.
Why else would we lose smell?
Here, again the evidence from post-mortem studies have provided refutation direct neuronal damage as a cause of anosmia, and instead peripheral cells such as substancellular cells within the nose are instead targeted.
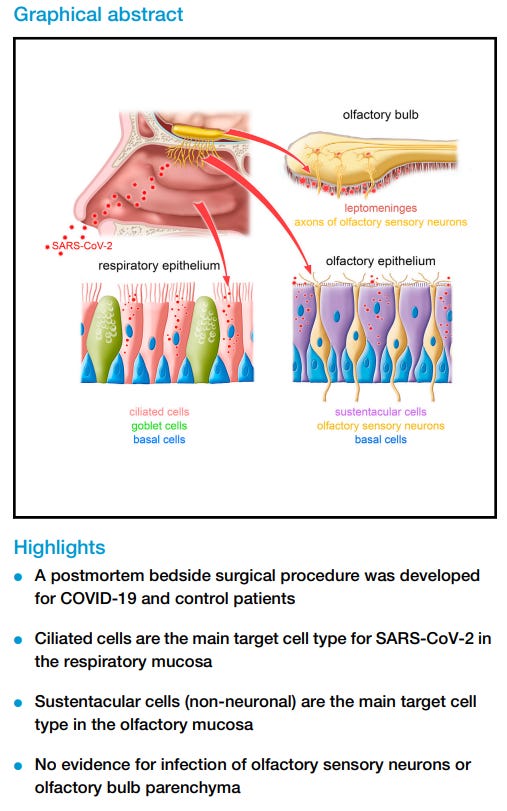
A study published in late 2021 from Khan, et al.3 took bedside endoscopic tissue samples immediately after the death of an individual who either died with or from COVID in an attempt to see what SARS-COV2 actually dose in the nose and olfactory bulb of infected individuals.
This study is rather detailed, and is one of the only studies that appears to have taken samples immediately after a patient had died.
So did this study show damage to the olfactory bulb and neurons within the nose?
No- instead, cells associated with olfactory mucosa, sustentacular cells, appear to have been the target of the virus, and it was infection and dysfunction of these cells that appear to have influenced the loss of smell.
Sustentacular cells are considered to be supportive cells, providing help to neighboring cells including olfactory neurons via metabolic aid and other cellular processes.
It appears that destruction of these cells are the main drivers of loss of smell:
We have here taken a virocentric view of COVID-19, from the viewpoint of SARS-CoV-2 acutely attacking the human olfactory system. We identified sustentacular cells as the main target cell type in the OM, failed to find evidence for infection of OSNs and of the OB parenchyma, and discovered viral RNA in the leptomeningeal layers surrounding the OB.
This evidence was supported even during the early months of the pandemic, and was noted in a July 2020 review when researchers noted that olfactory neurons don’t contain the proper receptors for SARS-COV2 binding.
Again, non-neuronal cells, more importantly sustentacular cells, are the target of the virus. The article cited by Harvard Medical School appears to be an in vitro study (the link for the study does not work), so one can argue that the Khan, et al. piece acts as confirmation for the in vitro results.
Sustentacular cells are constantly replaced, and so it’s possible that the return of smell is contingent upon the ability to replace destroyed cells.
But again, the evidence points to a refutation of previously held ideas that SARS-COV2 directly targets the neurons, and that the brain of those infected are being directly harmed from the virus.
Why the inconsistency and continuously-held belief?
So why exactly is it still argued that COVID may directly target the brain and other aspects of the nervous system, or why is it that results from studies are inconsistent?
Tissue sampling runs into a serious issue of cross-contamination, and so collection of samples may result in the spread of viral proteins during collection that would otherwise not be there. There’s also the fact that many staining techniques, as outlined in the previous post, run into an issue of cross-reacting and binding to other proteins and may create false positives.
Autopsy and post-mortem samples are done after the disease has run its course. Thus, the finding of viral proteins or RNA does not provide an explanation for the pathological nature of these proteins (the bystander effect).
Animal models, which have been used extensively as representations for what may happen in humans, are not actually good comparisons, even when monkeys are primates are used. Most studies in animals may not translate over well for humans, as is the case for many therapeutic trials that attempt to deliver drugs into the CNS of humans. Animal studies may show resounding success but then find no delivery in humans. Given that most evidence of neuroinvasion has relied on in vitro, in silico, and animal models, it’s possible that false assumptions were made about how dangerous the virus is when relying on models that don’t corroborate to evidence in humans, as reported in post-mortem and autopsy reports.
But probably more importantly is the fact that brain damage from SARS-COV2 just makes for good fear porn.
Recently Bret and Heather discussed an article from El gato malo in which questions were raised towards the role that over-prescription of aspirin may have had in the worse outcome of patients during the Spanish Flu (timestamped below).
It raises questions as to whether intentional malfeasance and practice of medicine may have worsened the perception of the Spanish Flu.
One can remember the early days of the pandemic and the images of people keeling over, the overfilled ER rooms and remember the fear that plagued much of the world. Even still, comments on long COVID are used as a evidence of some sort of brain damage.
Thus, what better way to instill that fear than to continuously remind people that their brain fog and loss of smell may be a consequence of permanent damage to their nerves?
What about the possibility of neurodegenerative diseases?
Here, the question still remains about the possibility that SARS-COV2 may exacerbate symptoms of Alzheimer’s Parkinson’s, and possibly prion disease. These same ideas and the constant search for amyloid are also reflected in fears over the vaccine.
One consideration is the prolonged neuroinflammation that may come from microglia activation and endothelial dysfunction brought on by the spike protein, rather than the spike protein directly leading to the misfolded proteins.
Microglia are essentially the innate immune arm of the central nervous system, and the actual role of microglia in neurodegeneration has been highly conflicted.4
However, similar to all manners of chronic disease continuous, prolonged inflammation have been associated with neurodegeneration(Streit, et al.5):
Neurodegenerative diseases – particularly Alzheimer's disease, but also amyotrophic lateral sclerosis, Parkinson's disease, and Huntington's disease – lack the prominent infiltrates of blood-derived mononuclear cells that characterize autoimmune diseases. On the other hand, there is abundant evidence that many substances involved in the promotion of inflammatory processes are present in the CNS of patients with such neurodegenerative diseases. By far the bulk of this body of evidence is related to studies in Alzheimer's disease [18]. What distinguishes Alzheimer's disease from other neurodegenerative diseases is the conspicuous presence of extracellular deposits of amyloid in senile plaques. Senile plaques in Alzheimer brain are present in different stages of maturity, ranging from diffuse to neuritic to dense core, but they all contain the amyloid beta protein (Aβ). Aβ is a peptide that forms insoluble and pathological extracellular aggregates that seem to attract microglial cells, as suggested by the clustering of microglia at sites of Aβ deposition (see [19] for a review). There is evidence from experimental studies in animals to support the idea that microglia can phagocytose and degrade amyloid [20, 21], but such phagocytosis is apparently either ineffective or inadequate in Alzheimer's disease. A key question within the current context is: "Does the amyloid in Alzheimer brain by itself represent a persistent injurious stimulus that causes neuronal injury, or are additional factors involved in eliciting this outcome?"
In prior discussion about immunotherapy for Alzheimer’s I’ve continuously raised the notion that researchers are unsure if the plaques from Alzheimer’s are directly involved with the pathology of the disease, or if they are a consequence of cellular dysfunction and the inability to maintain clearance of misfolded proteins— again, an issue of contextualizing the bystander effect. If therapeutics target the removal of plaques they may not be doing much to help with the actual progression of the disease if the plaques themselves are not directly involved. This may explain the highly contentious findings of breakthrough treatments in the field of Alzheimer’s.
What’s likely happening with SARS-COV2 and neurodegeneration is one idea raised by Nuovo, et al., in which it’s possible that microglia already stressed may not be able to handle the additional stress brought on by the endothelial dysfunction.
This may explain the worsening of symptoms in those with neurodegeneration already, but it also may lead to an unmasking of neurodegenerative disease in those predisposed to these diseases later in life.
That is to say, new-onset neurodegenerative diseases may be occurring in those who may be more at risk of these diseases in the future, whether due to environmental circumstances or genetic predisposition.
Thus, one can wonder if the reports of prion diseases related to the vaccines may be occurring in those who are more likely to experience these diseases in the future. Genetic testing and other examinations may provide some insights, but like with everything related to the vaccines it doesn’t appear much research is being done to consider these possibilities.
In that regard, I suppose Dr. Tanzi should be more worried about a family history of neurodegeneration putting him more at risk of Alzheimer’s post-infection than the virus alone.
Spike presentation is not the same between vaccine and infection
The last words I’ll discuss is one idea worth considering, and one that many have already raised since the rollout of the vaccines.
It appears that in the progression of SARS-COV2 the main damage done to peripheral organs such as the brain and heart are due to circulation of spike from the lungs rather than the virus itself. Whether this is brought on after substantial damage has already occurred, or if transport of spike may be mediated by exosomes, one can argue that circulation of spike from the lungs are likely to be heavily attenuated until the disease progresses substantially, in which case the barrier between the lungs and the bloodstream are compromised allowing free exchange.
This may explain why regions such as toes, fingers, the brain, and even genitals may be influenced by spike due to the microvasculature within these regions who may be more susceptible to even small doses of spike.
So in that regard the lungs may serve as a buffer for spike within the blood.
But can the same be said for the vaccines?
Irrespective of concerns over intramuscular vs intravenous injection, injection of the vaccines is providing an immediate, substantial payload of spike, and likely one that generally occurs over the course of days in those who are naturally infected.
This should raise concerns about the greater risk of spike circulating, and it’s likely that the inability to quickly sponge up spike due to a lack of adaptive immunity may explain many of the adverse reactions. This may also explain why, as reported anecdotally, that those who were naturally infected may have lower reported cases of adverse reactions, and in those who do other mechanisms may be at play.
More on this last point will be explained in a series sometime in the future.
Substack is my main source of income and all support helps to support me in my daily life. If you enjoyed this post and other works please consider supporting me through a paid Substack subscription or through my Ko-fi. Any bit helps, and it encourages independent creators and journalists such as myself to provide work outside of the mainstream narrative.

Nuovo, G. J., Suster, D., Sawant, D., Mishra, A., Michaille, J. J., & Tili, E. (2022). The amplification of CNS damage in Alzheimer's disease due to SARS-CoV2 infection. Annals of diagnostic pathology, 61, 152057. https://doi.org/10.1016/j.anndiagpath.2022.152057
Matschke, J., Lütgehetmann, M., Hagel, C., Sperhake, J. P., Schröder, A. S., Edler, C., Mushumba, H., Fitzek, A., Allweiss, L., Dandri, M., Dottermusch, M., Heinemann, A., Pfefferle, S., Schwabenland, M., Sumner Magruder, D., Bonn, S., Prinz, M., Gerloff, C., Püschel, K., Krasemann, S., … Glatzel, M. (2020). Neuropathology of patients with COVID-19 in Germany: a post-mortem case series. The Lancet. Neurology, 19(11), 919–929. https://doi.org/10.1016/S1474-4422(20)30308-2
Khan, M., Yoo, S. J., Clijsters, M., Backaert, W., Vanstapel, A., Speleman, K., Lietaer, C., Choi, S., Hether, T. D., Marcelis, L., Nam, A., Pan, L., Reeves, J. W., Van Bulck, P., Zhou, H., Bourgeois, M., Debaveye, Y., De Munter, P., Gunst, J., Jorissen, M., … Van Gerven, L. (2021). Visualizing in deceased COVID-19 patients how SARS-CoV-2 attacks the respiratory and olfactory mucosae but spares the olfactory bulb. Cell, 184(24), 5932–5949.e15. https://doi.org/10.1016/j.cell.2021.10.027
Peggion, C., Stella, R., Lorenzon, P., Spisni, E., Bertoli, A., & Massimino, M. L. (2020). Microglia in Prion Diseases: Angels or Demons?. International journal of molecular sciences, 21(20), 7765. https://doi.org/10.3390/ijms21207765
Streit, W.J., Mrak, R.E. & Griffin, W.S.T. Microglia and neuroinflammation: a pathological perspective. J Neuroinflammation 1, 14 (2004). https://doi.org/10.1186/1742-2094-1-14




I’m still in the middle of reading this but it’s really taken my interest. My grandmother (memere) and dad both died of neurodegenerative diseases. Alzheimer’s and and in my dads case- Alzheimer’s but suspected Lewy Body. I have had brain fog and intermittent short term memory loss since COVID as well as loss of smell. I’ve wondered and wondered what, if any the link could be with these disorders and covid. This is immensely interesting! Thanks
Shocker! It’s a respiratory virus not at all unlike all the others which came before it and its “novel” features appear to be a result of observation / confirmation bias.
We learn nothing do we? The past few decades have seen several viruses causing “absolutely characteristic” clinical syndromes - until it transpired they actually hadn’t.
Th