About Original Antigenic Sin...
Part II: Correction to Previous UK Post, Applying it to SARS-COV2, and Concluding Remarks
Piecing it Together- Is Original Antigenic Sin Occurring?
Adding Context to my Previous Position
In my previous post I did not allude to the idea of Original Antigenic Sin, and to be frank I didn’t know anything about it at the time.
Instead, I argued that viral clearance from previous antibodies may prevent the immune system from being exposed to the N protein and thus an immune response was shunted because the N protein could not be recognized.
I used this as justification for the note indicated in the UK Surveillance Data, however I should clarify, and possibly correct, this position.
There is actual evidence of the Nucleocapsid protein escaping the infected host cell after lysis. In fact, the nucleocapsid protein may be responsible for inducing a pro-inflammatory response in the body, as indicated by Gao et. al. 2020:

And there is evidence that anti-N antibodies may be produced earlier than spike protein antibodies, possibly because of its ability to induce inflammation and cause damage, although this idea should be taken with skepticism.
So instead I’ll argue that the situation is complex, and unfortunately my original justification may not be enough to argue what we are seeing. There is evidence to justify that if the anti-S antibodies are able to mount an immune response before the body can recognize the N protein it may lead to viral clearance without producing a strong Anti-N antibody response. We can also add weight to this claim by noting that less severe infections usually lead to less robust antibody responses. There is also evidence to suggest that those who have been vaccinated have reduced symptom duration, and we can conjecture that their reduced sickness duration may reduce the ability to produce anti-N antibodies, leading to what we are seeing in the UK Surveillance Data.
Nonetheless, the situation is far more complex than I previously alluded to, and this needs to be taken into account.
This also means that people who have alluded to the possibility of OAS without examining the previous studies and data are also missing out on the complexities and nuance of the situation, because OAS, if applied, does not operate within this scenario alone.
Applying OAS to anti-S Antibodies
If we are to evoke OAS, we need to contextualize it to all manners of what we are examining. This means that we can’t just look at it in the context of SARS-COV2 vaccines and recent exposure, but one’s entire infection history.
The UK data indicates strong anti-S titers, and we may be able to explain this with OAS. We know that previous exposure to SARS-COV creates cross-reactive antibodies to SARS-COV2. In fact, there is evidence that even exposure to seasonal coronaviruses elicited cross reactive antibodies to both seasonal coronaviruses and SARS-COV2 spike proteins.
So how does this work with OAS?
Well, this would indicate that vaccination with a vaccine that targets the spike protein, as we are doing with SARS-COV2, may not be producing a specific antibody response to SARS-COV2’s spike protein, and may instead be engaging in “back boosting”- anti-S antibodies against seasonal coronaviruses may be activated instead of naive B cells that would target the spike protein of SARS-COV2 specifically. This fits with the ideas outlined by Smith et. al. 1999 and Monto et. al. 2017 of “negative interference”- the response to antigen-based vaccines will be interfered with by the body wanting to mount previously learned anti-spike antibodies.
This may also indicate a confounding variable about why we may not be seeing strong neutralizing abilities against Delta with the vaccine; it may not be that the vaccines were produced using the Wildtype (Wuhan) variant spike, but that previous exposure to human coronavirus spikes is causing OAS to occur and masking the immune system’s ability to properly respond to the vaccine, and instead may cause it to rely on previous memory B cells’ antibodies to target the vaccines. If Wildtype spike can be cleared by these cross-reactive antibodies, a proper antibody response against the Wildtype spike will not be produced, and instead antibodies against previous human coronaviruses will be strengthened.
So the strong anti-S antibody levels may not correlate with the vaccine or SARS-COV2 itself. Note that this is highly speculative (we do not even know if OAS is occurring), but this highlights the point that in order to evoke OAS we need to examine all possible instances of OAS occurring, and not where we want to see it (as some people have done). In some sense, think of Chesterton’s Fence (er, Chesterton’s Original Antigenic Sin?)
OAS in the case of Vaccines
Before we dive into the OAS and Vaccines I want to clarify something that I continue to see being misinterpreted.
There’s a lot of misconceptions around the use of Spike protein as a vaccine target. Unfortunately, we now know that the spike protein is cytotoxic, and it seems that this was not noticed during vaccine development. The reason why vaccine development targeted the spike protein specifically, is that there were hopes that it would lead to neutralizing antibodies.
Here, once again, people misinterpret the notion of neutralizing antibodies. The intent of neutralizing antibodies is that they must neutralize infection. That means that their mechanisms of action must prevent the virus from entering into the host cell, and the best way to do that is to target the antigen that binds to host cell receptors. In this case, it is the spike protein. The Nucleocapsid protein cannot act as a target for neutralizing antibodies because it is hidden away within the virus. Yes, it does appear to be able to spread outwards from the body, as indicated by the Gao et. al. Study, but that occurs AFTER viral replication and translation; viral binding via the spike protein precedes the production of Nucleocapsid proteins.
We do see strong and early antibody titers against N-proteins over the course of an infection. However, because the N protein is sequestered within the virus, the development of a vaccine against the Nucleocapsid protein would be much less effective since it would require the immune system to notice the protein in order to mount a response. Therefore, in theory, the spike protein was the best possible candidate to utilize in developing an antigen-specific vaccine (although we are seeing the possible issues now).
Also, if you would like more information on the spike protein, I highly suggest you all take a look at Joomi Kim’s Substack piece from “Let’s be Clear”:
There is some evidence of OAS occurring with flu vaccines (remember this is still controversial and studies examined inactivated viruses), and we can infer the issues that may arise with our vaccination procedures as we have it now.
Almost all vaccines that are available in the US or in development target the spike protein specifically.
The argument being made by some is that the mRNA vaccines should be updated to utilize Delta’s spike protein, but if we evoke OAS we can see the large issue here.
Forgoing previous exposure to human coronaviruses, we can see that prior vaccination with the Wildtype spike may cause “back-boosting” of antibodies against Wildtype spike instead of creating a new immune response against Delta-spike. If this were to occur, vaccination with a different spike protein may be futile; it’ll lead to the same situation as the Gardasil and Gardasil 9 predicament. The antigenic distance between Wildtype and Delta’s spike protein is small, and therefore negative interference should be expected to occur (this would also fit with Brown et. al.’s (B) scenario).
However, vaccination with a different antigen should not have the same effect. If vaccines were administered that targeted the Nucleocapsid protein specifically, the negative interference from a previous spike-specific vaccine should not occur; they are two completely different antigens.
So instead of creating new vaccines targeted towards Delta’s spike protein, a vaccine that targets other antigens should be considered if OAS is a concern.
But what if a vaccine was produced that targeted both the spike protein and the nucleocapsid protein? Should we expect OAS to occur?
Let’s speculate on this position, and when we do let’s note that there is something we need to get rid of when examining this scenario. We have to understand that even though we ascribe behaviors to the immune system that make them seem “cognitively aware” this may not actually be occurring; our immune systems may not be able to relate the information that any antigen it notices was derived from [this] pathogen and that both [this] antigen and [that] antigen come from the same pathogen.
So in this scenario, let’s assume that someone was previously vaccinated and produces anti-S antibodies. Now, they are given a vaccine that contains both the Nucleocapsid and Spike protein.
Our prejudgment, due to us being cognitively aware, may try to piece together the idea that both are derived from SARS-COV2, this person has antibodies against the spike (and thus SARS-COV2), and if we apply OAS that means that the immune system may spend all of its time targeting the Spike protein and not target the Nucleocapsid protein.
Remember that, for all intents and purposes, the immune system does not know that the Spike and Nucleocapsid proteins are derived from the same pathogen; it only knows that is has had previous exposure to one antigen and not the other.
In this case it would be hard for us to argue that somehow the immune response against the Nucleocapsid protein would be hindered by the previous exposure to the spike protein; we’d be in serious trouble with the vast amounts of pathogens we come into contact with daily if that were the case.
Therefore, I would suspect, although we’ll need evidence, that this type of vaccine may not cause any possible OAS to occur.
Edit: Adenovirus Vaccines
One of the areas with the most concern in regards to OAS would be the adenovirus vaccines.
The adenovirus-based vaccines utilize either a chimp or a recombinant vector. If a human adenovirus vector was used there would be concern that in areas with high adenovirus circulation that these people would mount an immune response and target the vector before it can elicit an immune response against the spike protein.
This would definitely be one of the scenarios to be concerned about when it comes to OAS, and it’s most likely one of the reasons why these vaccines were only meant to be one dose.
Brown et. al.’s Hypothesis and what we are Seeing
By now I would argue that there are faults with the proposed (B) hypothesis. I find it difficult to imagine that completely separate antigens should be affected by the previous exposure to the other antigen. Granted, I could be wrong (really, all of this could be purely BS), but based on what I’ve outlined this does not seem like a likely case.
Therefore, I would be hard pressed to argue that what we are seeing exactly alludes to OAS. This also indicates that vaccinations with whole, inactivated viruses may not be a concern with respect to OAS, even in someone who was previously vaccinated with the Wildtype Spike protein alone, although we would need to see studies to conclude that to be the case.
And what about the UK data?
The UK data does indicate high levels of S antibodies and relatively low levels of N antibodies. However, we can still make some sense of the data.
Note that the graph for the N antibody titers is not “flat”. Instead, I would argue that there seems to be an age-related dependency on the curve’s graph. Younger people have much higher anti-N antibody titers, and they indicate that they increase over the course of time indicated.
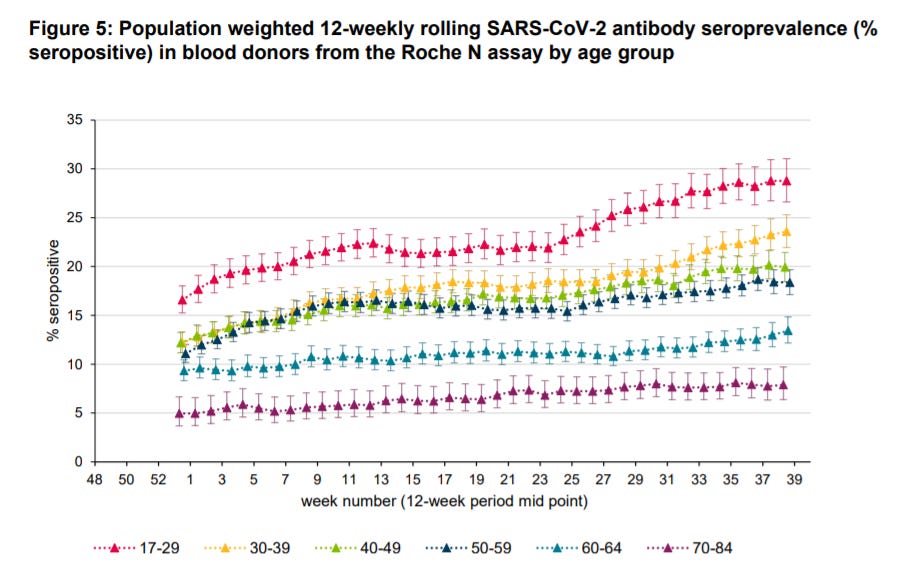
Also note that in the younger demographics there is almost a doubling of anti-N antibodies (the red and yellow lines). So instead of arguing that OAS may be occurring, it may be better to assume that we may be noticing immune senescence at play here instead; younger people are overall able to produce a robust antibody response to infections compared to the elderly.
But then what about this graph that looked at anti-S antibody titers? Can we be sure that immune senescence is at play here?

One thing that people absolutely seemed to have overlooked is the y-axis of this graph. We can see here that before vaccination it seems like both anti-S and anti-N antibody titers are relatively similar (~10%). Then, after vaccination, there’s a large jump in the Roche S levels.
What’s important to take away here is that the y-axis has been stretched in order to accommodate the large spike in Roche S. The Roche N line may look “relatively flat” but note that the starting point was around 10% seroprevalence. Where is it at the 40 week mark? Around 20%, indicating a doubling of anti-N antibody titers.
So here we can see that people may have been played by statistical trickery. I bet if you separated the 2 lines and made one specifically for the Roche N you will absolutely notice a large increase.
Now, we have no way of knowing if this related to the increase in infection, and we have no way of knowing what can be attributed to people who have been vaccinated and then infected. Also, just like I stated with the Roche N graph specifically, we’d have to argue whether or not immune senescence is at play for the elderly- we do see that the lines for the younger demographics match the red Roche N line seen in the antibody seroprevalence graph, but I’ll leave that to more statistically savvy people to figure out.
Unfortunately, any possible line for Roche S antibodies between vaccinated, infected, and vaccinated & infected are hard to distinguish from this dataset alone.
But what remains clear is that there really is no way of knowing whether or not OAS is at play here just from the data, and it doesn’t help when people misinterpret the data just by looking at the lines and examining the graphs.
It also doesn’t help that the argument in point (iii) that everyone has been referencing is EXTREMELY general. It doesn’t take into account age or severity of illness, all of which I have indicated play strong roles in immune response.
Concluding Remarks
I hope by now you notice how much of a clusterfuck all of this really is (seriously, welcome to complex systems!)
Even then, I hope this highlights how we need to approach all of this with a nuanced, grounded approach.
So in this instance I will say that my previous post holds merit; maybe priming of the Spike protein is causing clearance of the virus before the body can produce an immune response against the Nucleocapsid protein. But we will need to find more evidence to support this idea, and really everything we are seeing.
But most importantly I need to reiterate the point that we do ourselves no favors by engaging in histrionic paranoia. There are absolutely valid reasons to question the vaccines and everything that is going on, but it needs to be approached with respect to the scientific process, and when we question the science we need to engage in scientific discourse and not hysterical suppositions.
I’ll leave this post with some things to consider that may help us find some answers. But note these are hypothesis and may not even be possible:
If we want to see if OAS plays a role, we may need to compare antibodies in people who have had previous hCOV exposure but not exposure to SARS-COV2 or the vaccine. Compare the antibodies with those from the vaccines and see if they overlap.
If OAS is a concern in regards to vaccines, and there is at least some evidence from flu vaccines, we need to be very cautious about what types of vaccines we administer, especially in those previously vaccinated with the Wildtype spike protein. People who have previously been vaccinated with the Wildtype spike are far more likely to experience OAS when given the spike of a different variant.
Vaccination may not be priming specific immune responses SARS-COV2, and may instead be engaging in “back-boosting” of previous immune responses to seasonal human Coronaviruses. Studies should look into this possibility.
Anti-N antibody titers should be examined for people who have been vaccinated but subsequently hospitalized by COVID infection. This will help to indicate if the relatively lower Roche N levels may be due to shorter viral infection (and thus quicker viral clearance) or due to possible OAS interfering with antibody production against the N protein. Variables such as age and immunocompromised status should be accounted for, as those may be more likely to indicate immune senescence or immune dysfunction than actualy OAS.
The last point (and possibly the most important): When discussing Original Antigen Sin and other hypothesis similar to this it’s important to argue in parallels and relativism. Compare similar antigens to antigens, vaccines to vaccines, and pathogens to similar pathogens.
There may be plenty more reasons, and if any of you have some please discuss in the comments below.
But for right now I just want to take a break and watch some horror movies.
Thank you for reading my newsletter. If you enjoy my articles please consider becoming a free subscriber in order to receive notifications.
And share with others who may find these newsletters interesting.
Also, please consider becoming a paid member. The research and work put into these articles takes many hours and being a paid subscriber allows me to continue to do this full time.
In-Text Citations
Brown et. al. 2021. Original Antigenic Sin: the Downside of Immunological Memory and Implications for COVID-19. Taken from https://journals.asm.org/doi/10.1128/mSphere.00056-21#B5
Smith et. al. 1999. Variable efficacy of repeated annual influenza vaccination. Taken from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC24180/
Monto et. al. 2017. The Doctrine of Original Antigenic Sin: Separating Good From Evil . Taken from https://academic.oup.com/jid/article/215/12/1782/3112608
Kim et. al. 2009. Original Antigenic Sin Responses to Influenza Viruses. Taken from https://www.jimmunol.org/content/183/5/3294.full
Gao et. al. 2020. Highly pathogenic coronavirus N protein aggravates lung injury by MASP-2-mediated complement over-activation. Taken from https://www.medrxiv.org/content/10.1101/2020.03.29.20041962v3.full-text
Long et. al. 2020. Antibody responses to SARS-CoV-2 in patients with COVID-19. Taken from https://www.nature.com/articles/s41591-020-0897-1
Guo et. al. 2021. Cross-reactive antibody against human coronavirus OC43 spike protein correlates with disease severity in COVID-19 patients: a retrospective study. Taken from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8023607/
Antigenic Seniority: Here’s another idea similar to Original Antigenic Sin that may be of interest. Both hypothesis lead to the same conclusions, albeit are a little different.
Lessler et. al. 2012. Evidence for Antigenic Seniority in Influenza A (H3N2) Antibody Responses in Southern China. Taken from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3400560/
Additional Sources:
Kim et. al. 2018. Influenza Virus: Dealing with a Drifting and Shifting Pathogen. Taken from https://www.liebertpub.com/doi/10.1089/vim.2017.0141
Coughlan et. al. 2018. Overcoming Barriers in the Path to a Universal Influenza Virus Vaccine. Taken from https://www.cell.com/cell-host-microbe/fulltext/S1931-3128(18)30328-7?_returnURL=https%3A%2F%2Flinkinghub.elsevier.com%2Fretrieve%2Fpii%2FS1931312818303287%3Fshowall%3Dtrue
Stamper et. al. 2018. What Are the Primary Limitations in B-Cell Affinity Maturation, and How Much Affinity Maturation Can We Drive with Vaccination? Taken from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5899041/
Gatto et. al. 2010. The Germinal Center Reaction. https://www.jacionline.org/article/S0091-6749(10)01416-8/fulltext
Noori et. al. 2021. “Original antigenic sin”: A potential threat beyond the development of booster vaccination against novel SARS-CoV-2 variants. Taken from https://www.cambridge.org/core/journals/infection-control-and-hospital-epidemiology/article/original-antigenic-sin-a-potential-threat-beyond-the-development-of-booster-vaccination-against-novel-sarscov2-variants/C8F4B9BE9E77EB566C71E98553579506




If you look at vaccine uptake by age group vs N prevalence by age you will also see a correlation.
I completely forgot to add: In the wake of all of this talk about boosters there should be serious concern about OAS occurring with the Adenovirus based vaccines. The vectors used are Chimp/recombinant derived adenoviruses because there is concern that using human adenovirus vectors in populations where adenoviruses are common may prevent proper vaccination due to the immune system targeting/eliminating the vector before it can induce spike protein formation.
It would make sense then that using a 2nd dose of one of the adenovirus vectors would prove very ineffective due to OAS and prior immune memory.