The Case of the Leaky Chickens
What can leaky vaccine studies in commercial chickens tell us about leaky COVID vaccines?
There’s no denying that the current COVID vaccines are leaky. They don’t stop viral transmission and there are questions as to how well they are reducing the severity of COVID in the recipients.
Considering that hundreds of millions of people worldwide have been provided these vaccines, several people have raised questions as to whether leaky vaccines may lead to the eventual emergence of more virulent strains of COVID.
Most evidence in support of these remarks come from studies of commercial chicken vaccination against Marek’s disease virus (MDV), and it appears that these chicken studies have actually been used as an argument in support of the leaky vaccine hypothesis.
Even Joe Rogan himself brought this up on one of his podcasts last year, referring to the highly influential study by Read, et. al.1 in regards to leaky vaccine concerns. However, it's worth noting that the lead author of the paper quickly came out and responded to Joe Rogan’s claims and argued in support of COVID vaccination.
As someone concerned with the truth and the parsing of information, it’s important to actually examine these studies and understand whether there is truth and validity in these concerns, or if there’s several caveats worth examining.
Transmission/Virulence Trade-off Hypothesis
Before looking at the actual study we need to take a look at the hypothesis that serves as the basis for said study.
You probably never put a name to this hypothesis, but you’ve likely heard of the argument that viruses exhibit some form of evolutionary trade-off. In viruses that are highly transmissible, they tend not to be very deadly: many people get infected but not many people die.
However, in highly virulent viruses the transmissibility tends to be rather low because the virus supposedly kills off far too many people before it can spread to others.
This trade off is one that’s derived from evolutionary theory and is informally named the transmission/virulence trade-off hypothesis2, and it suggests that a virus must balance both high transmission and high virulence in order to properly fit the population that the virus traditionally infects.
For example, let’s say that a virus infects the entirety of a select population, yet those people don’t die. The virus continues to circulate within this select population, and over time the virus begins to mutate, has higher tropism for different organs, or has higher binding affinity to our cells. The virus now begins to become more invasive and more damaging to our cells. However, younger and healthier people within this population tend to do well with this virus, but the elderly and immunocompromised do not. Thus, a high level of the elderly die off from this mutated virus.
At the point that a large subset of the population begins to die off is when a “signal” is sent out that essentially says this is as far as a virus can mutate before the virus kills itself off along with its host. This threshold essentially puts a hypothetical cap on a virus’ virulence, and at this point the cost for virulence is far too high to continue down the path of greater virulence.
There are a few additional explanations for this trade-off, such that more virulence would lead to more severe symptoms and more severe symptoms prevent people from interacting with others to a greater degree. Therefore, more crippling infections would immediately halt social interactions and the exchange of more virulent pathogens along with it.
In order to explain how this relates to leaky vaccines, let’s start with the hypothesis that vaccines should reduce the risk of death. Essentially, vaccines should alter the threshold of virulence since survivability would be altered to benefit us. If this change in threshold was achieved with perfect (all-or-nothing) vaccines then that would be the end of the discussion. However, with leaky vaccines in which a virus can still be transmitted, the change in virulence threshold means that those who would have otherwise died have will now be spared. The fitness signal is altered, and a virus may now mutate and become more virulent to now kill off this spared population.
Overall, this hypothesis has taken on a life of its own and even many laypeople who don’t have knowledge of virology or immunology have discussed this hypothesis. Later on we will see why there are many problems with this hypothesis, and whether there is actually any evidence to support the veracity of the hypothesis as well.
The Leaky Chicken Study
With that bit of information about this transmission/virulence trade-off hypothesis we can now turn to the leaky chicken study.
This study titled, “Imperfect Vaccination Can Enhance the Transmission of Highly Virulent Pathogens” was conducted in 2015 and was based off of prior assumptions that leaky vaccination among commercial chickens was responsible for the increased virulence of Marek’s disease among these chickens.
Prior to vaccination there were concerns that widespread infection among commercially-raised chickens would severely halt poultry production and damage the poultry industry. Thus, vaccines were used as a method to keep infection rates down and production yield high.
One of these diseases of concern was Marek’s disease virus (MDV), which is caused by a herpesvirus. Earlier strains of the virus usually led to relatively mild symptoms in most chickens with the older chickens usually suffering paralysis from infection. However recent, more virulent strains of MDV have been found to be oncogenic (cancer-forming) and have lead to formation of lymphomas in some of these infected commercial chickens:
Chickens become infected with MDV by inhalation of dust contaminated with virus shed from the feather follicles of infected birds. In a contaminated poultry house, chicks are infected soon after hatching and remain infectious for life [16]. The virus can remain infectious in poultry dust for many months [17,18]. As originally described, Marek’s disease (MD) was a paralysis of older birds, but by the 1950s, “acute MD” characterised by lymphomas in multiple organs in younger birds was occurring. This became the dominant form of MD, with increasing virulence, characterised by more severe lymphomas and mortality at increasingly early ages and, under some circumstances, paralysis and death in the first weeks of life, well before lymphoma formation [15,19].
So it’s the association between vaccination and the emerge of more virulence MDV that led the researchers to test out this transmission/virulence trade-off hypothesis. Do commercial chickens continue to spread Marek’s disease to one another, or does vaccination with even a leaky vaccine reduce transmission rates and provide some protection?
Because this study is broken down into a few sections I’ll cover each individually based on how the authors categorized the paper.
1. Viral Shedding
The researchers first wanted to see if both vaccinated and unvaccinated chickens would produce different rates of viral shedding. They also wanted to see if different virulent strains would alter viral shedding as well.
The vaccine used in this study is a live, serotype 3 herpesvirus of turkeys called HVT. It’s the most widely used vaccine in commercial chickens.
Here, the researchers used Rhode Island Red (RIR) chickens since they were not selectively bred and should be naïve to MDV- they should have no prior immunity. Chickens were separated into groups and either vaccinated (dark, solid lines and colors in graphs) or unvaccinated (light, dashed lines and colors in graphs) and were challenged with different strains of the herpesvirus.
As an aside, the researchers use terms such as “hot” and “cold” to refer to virulence, such that “hot” strains are more virulent while “cold” strains are relatively mild in nature.
These chickens were challenged and viral load was measured based on PCR testing from dust collected from the chicken’s isolators. The results can be seen in the chart below. Keep in mind that the color coding to differentiate the strains are done throughout the paper.
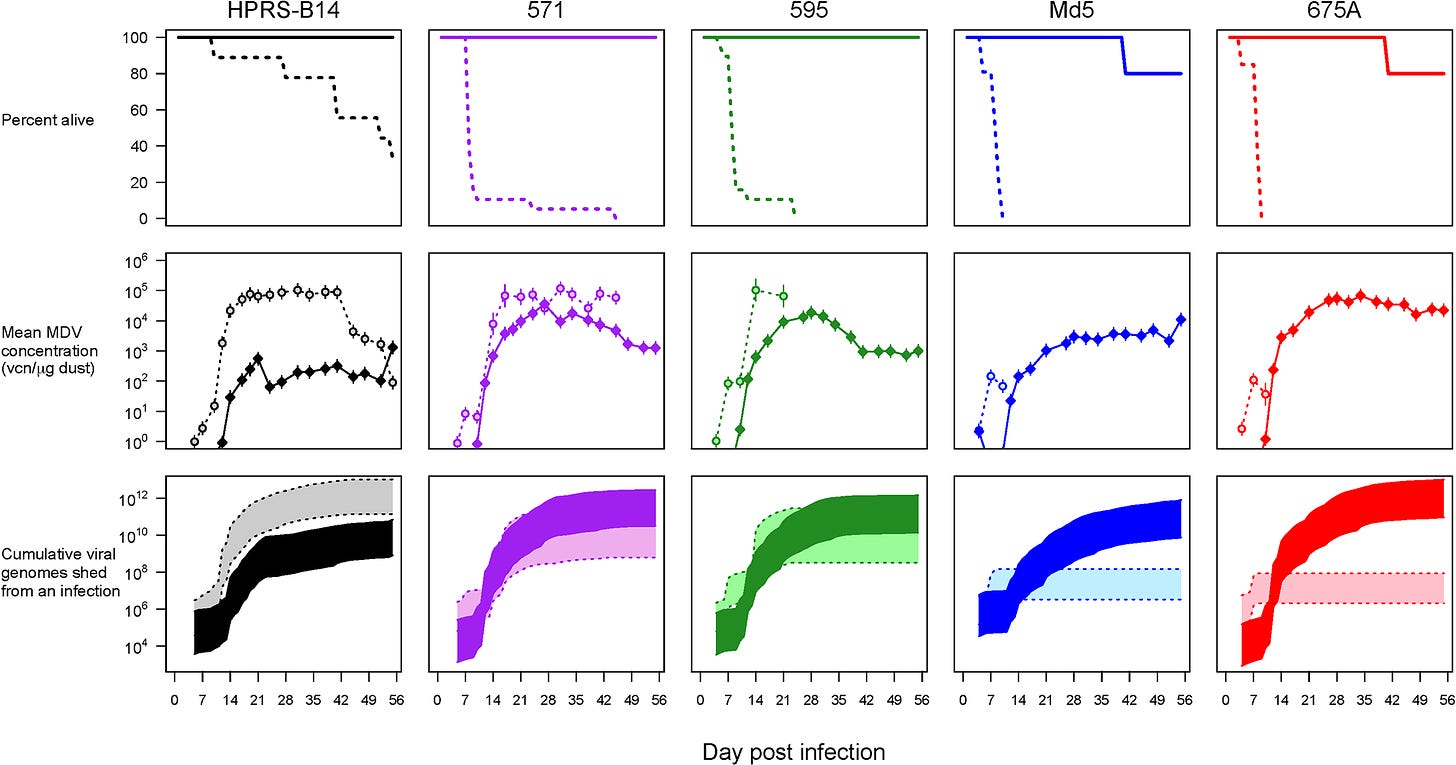
As expected, the researchers found that hotter strains of the virus led to very quick death among the unvaccinated chickens (first row, dotted line). Unvaccinated chickens also tended to have a higher rate of viral shedding (second row, dotted line).
However, the early death of the unvaccinated chickens with the hotter strains of the disease meant that they were not capable of shedding for extended periods. Instead, the vaccinated chickens continued to have persistent viral infection (note that this is from PCR testing) for several weeks with continuous viral shedding.
For the researchers these results validated the assumption that more virulent strains of the herpesvirus would kill infected individuals off before they can transmit their infection:
By preventing death, vaccination greatly increased the infectious period of the most virulent strains, increasing the total amount of virus shed by several orders of magnitude, and increasing it above that of the least virulent strain (Fig 1, bottom panels). Thus, the net effect of vaccination on both host survival rates and daily shedding rates was to vastly increase the amount of virus shed by virulent strains into the environment.
2. Onward Transmission
Experiment 1 of the study showed that vaccinated chickens may continue to shed the virus for several weeks, yet it didn’t show how this shedding would influence other chickens.
The next experiment wanted to test whether an infected chicken could infect and kill off chickens that were housed together.
Researchers took both vaccinated and unvaccinated chickens, inoculated them with the 3 most virulent strains of MDV, and housed these birds with immunologically naïve chickens called sentinels to see if the sentinels would become infected and die.
The results of Experiment 2 are shown below.
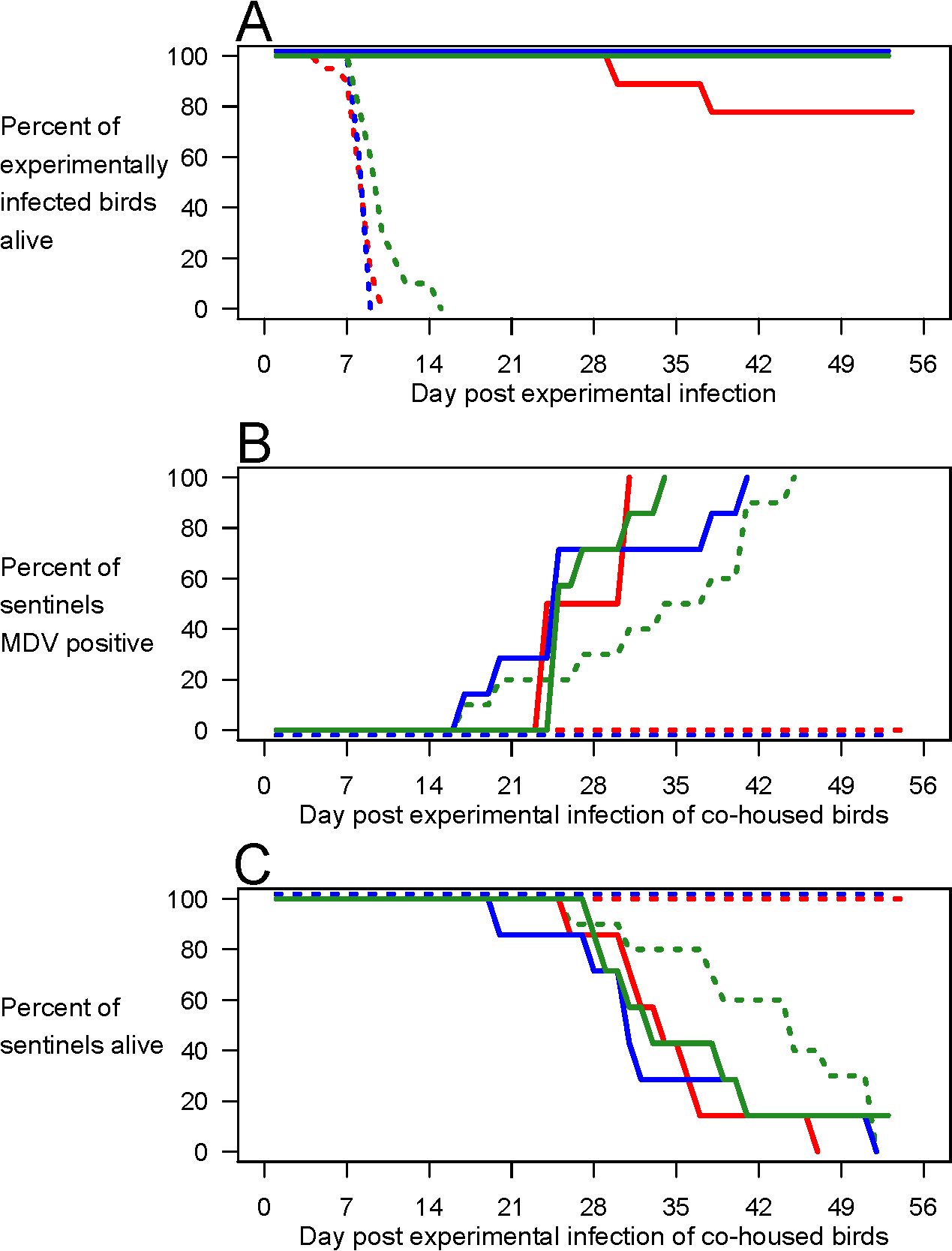
Interestingly, the unvaccinated chickens died off relatively quickly after becoming infected without engaging in viral shedding and infecting the sentinel chickens (dotted lines). However, the vaccinated chickens continued to exhibit viral shedding which eventually led to the death of the sentinels (bold lines), suggesting that vaccination led to persistent infection and extended shedding.
Thus, in accordance with the imperfect-vaccine hypothesis, vaccination enabled the onward transmission of viruses otherwise too lethal to transmit, putting unvaccinated individuals at great risk of severe disease and death.
The only strain to deviate from this relationship was the 595 strain (green line), and the researchers suggest that the reduced lethality may have allowed for a greater period of viral shedding in the unvaccinated chickens, leading to the eventual infection and death of the 595 sentinels.
Thus, slight reductions in lethality can be sufficient to allow onward transmission. Nonetheless, even for strain 595, vaccination led to more rapid infection of sentinels (Fig 2B; median time to positivity 9 d earlier than in unvaccinated birds, p < 0.05), thus increasing the rate at which secondary cases were generated, a critical determinant of both viral fitness and case incidence in a rising epidemic.
3. Transmission between Commercial Birds
Note: I'm skipping over the maternal antibody section since it’s not entirely necessary for this discussion (and I can see the “near email length” banner popping up at any moment). Just know that mother hens can pass antibodies onto their offspring in order to provide them with a bit of support as they make their way into the world. These antibodies protect against less virulent strains but not so much against the more virulent ones.
This brings us to the last experiment. The previous experiments suggest that, in certain settings, the transmission/virulent trade-off hypothesis was validated. However, these settings were not comparable to actual commercial broiler settings which would be one in which the more virulent Marek’s disease supposedly emerged from.
Our experiments above show that direct vaccination of birds or vaccination of parent hens makes possible the onward transmission of viral strains otherwise too lethal to transmit, and thus that unvaccinated individuals are put at increased risk of severe disease and death. However, in a modern commercial broiler setting, all birds in a flock would originate from vaccinated hens (and so would be positive for maternally derived antibody), and also be vaccinated. We thus set out to determine whether our most virulent strain could transmit to vaccinated sentinels, a necessary condition for persistence of hyperpathogenic strains in the modern industry (Experiment 4).
Thus, this was the last setting that the researchers looked into. The researchers took chickens from mothers who were previously vaccinated (implying maternal antibodies passed onto chicks) and subjected them to HVT vaccination. These chickens were then inoculated with the most virulent strain in this study (675A- in red) and cohoused with other sentinels that were either vaccinated (solid lines) or unvaccinated (dotted lines). The study was conducted twice (red vs blue lines) and the results are shown below.

Under all circumstances sentinels became infected with MVD and were shown to have comparable viral shedding (A+B), with the main difference being that unvaccinated sentinels eventually died while vaccinated sentinels were spared from death (C+D).
Importantly, all sentinels, vaccinated and unvaccinated, became virus positive in the feather follicles, meaning that they themselves started shedding. Vaccination protected sentinel birds from death (Fig 4C), prolonging infectious periods by about 2 wk (Fig 4D; standard error of the difference ±3.2 d, F1,36 = 19.9, p < 0.0001). Thus, not only does our most virulent strain transmit between modern commercial broilers when they are vaccinated, the duration of shedding in the next step in the transmission chain is also increased by vaccination.
Although with its own limitations, this final experiment at least indicates that in a commercial setting that imperfect vaccines can reduce death in vaccinated chickens while also transmitting the virus to nearby chickens and possibly killing those not adequately protected.
Providing Context to the Leakiness
So far this study from Read, et. al. has validated some of the concerns over imperfect vaccination. The vaccinated chickens could still infect other chickens, and in the case of the more virulent pathogens the chickens who were unvaccinated eventually died off.
The imperfect-vaccine hypothesis was suggested as an evolutionary mechanism by which immunization might drive MDV virulence evolution [2], but there has been no experimental confirmation. Our data provide that: by enhancing host survival but not preventing viral shedding, MDV vaccination of hens or offspring greatly prolongs the infectious periods of hyperpathogenic strains, and hence the amount of virus they shed into the environment.
But does this really relate to what we are seeing with these COVID vaccines, and is this study the be-all/end-all of leakiness?
Well, for one remember that this study didn’t actually measure the evolutionary flow from less virulent strains of the virus to the more virulent ones. The researchers even comment that they’re not sure themselves if vaccines led to the hyperpathogenic strains of MDV that killed off all of the unvaccinated chickens:
Our data do not demonstrate that vaccination was responsible for the evolution of hyperpathogenic strains of MDV, and we may never know for sure why they evolved in the first place. Clearly, many potentially relevant ecological pressures on virulence have changed with the intensification of the poultry industry.
So even though the researchers suggest that their experiments validate the imperfect vaccination hypothesis, there aren’t any causative explanation provided; at least from this study.
There’s also the fact that that commercial chickens began to present with lymphomas in the 1950s while the first commercial vaccination measures began during the 1970s, which would at least allude to increasing virulence of MVD even before the introduction of leaky vaccines. In fact, it is likely that the first vaccinations were done in order to combat the increasing virulence of MVD rather than cause the initial virulent strains, which would once again counter the assumption that leaky vaccines were the initial cause of the virulence. Instead, it could be that leaky vaccination allowed for maintenance of these viral strains. The actual evidence proposed from this leaky chicken study would be more fitting in arguing virulence maintenance rather than increasing evolutionary virulence.
It also doesn’t help that the transmissibility/virulence trade-off hypothesis is actually a highly contentious hypothesis. In fact, there is no actually connecting evidence to suggest that death should lead to less virulent pathogens. For example, take this excerpt from Bull, J. J. & Antia, R.3 which discusses the GMNR model (a model similar to the leaky vaccine hypothesis used throughout this article):
The GMNR model specifically requires the penalty for increased transmission to be increased host death (higher virulence), that a higher transmission rate would incur too many excess host deaths for the gain in transmission rate (see below for a consideration of a trade-off with recovery rate). This mortality assumption has been commonplace in modeling evolution of virulence [15, 17, 27–30]. It persists as a general assumption despite limited evidence [e.g. [31]] because there has been little direct evidence to the contrary, it seems plausible and it gives substance to the model predictions (change in deaths has tangible meaning). It is perhaps surprising that, for the vast majority of human viruses, we know almost nothing about what would limit higher transmission. We do know that host death and recovery halt transmission (for most infections). But the key question for evolution of virulence is what would reduce the number of net transmissions from a virus with a higher transmission rate. The fact that dead and recovered hosts do not transmit does not tell us what offsets the gain from a higher transmission rate—except to tell us that any increases in host death or recovery will work against net transmissions.
Additionally, one meta-analysis4 conducted to examine the relationship between virulence and transmission was not able to find any evidence, and instead found a high level of heterogeneity within the literature. This would suggest that the hypothesis may be very limited in scope rather than the broader, more generalized manner that we tend to use it.
So ironically, the hypothesis that many people use to explain viral evolution may not actually be substantiated by the literature or evolutionary principles. Its use may be highly specific and dependent upon the virus.
So how could we explain the possible increase in virulence of MVD?
Read, et. al. provides some ecological assumptions, but the outline from Bull, J. J. & Antia, R. dissected these factors very well and mimics some of the sentiments from Read, et. al. Essentially, the right factors were at play to create the perfect environment for increasing virulence:
We note that a key difference between MDV and many human vaccines is the very point of GMNR and Read et al. [1]—that the vaccine does not fully protect against infection and transmission. But as there are some human vaccines that allow re-infection, we need to explain why those do not lead to the MDV-like outcome. Thus MDV may be special:
The agricultural setting of chicken husbandry is ripe for the evolution of a virus with rapid infection. Birds are crowded with a short harvest time. In this environment, a high mortality rate has little cost to the virus, provided the virus achieves a high transmission rate while the host is alive [56–58].
MDV transmission in the chicken house does not require contact from a live bird, only that the infected bird shed virus onto the floor. This feature relaxes selection against killing the birds, provided infected birds produce infectious virus before they die [1, 56–58].
Vaccination of housed chickens is near 100% for many houses, so selection is more intense than if coverage was moderate (as is true for some human vaccines).
MDV is an alpha herpesvirus, a class of virus that typically establishes latent infections with lifetime transmission. MDV is specifically an oncogenic virus, causing tumor formation in its wild-type state [59]. Herpesviruses have many defenses against host immunity, which in turn enable the virus to persist within the host, and superinfection is known for many. This plethora of anti-immune responses may provide the virus with many pathways to evolve immune escape. T-cell responses are important defenses against at least some beta herpesviruses, and the genetic uniformity of chickens in the house may remove the variation in T-cell repertoires important in host population defense against MDV. Genetic uniformity is not expected to be a sufficient explanation for the evolution of hypervirulent MDV, as it should facilitate virulence evolution of other chicken pathogens, but it may be a contributor.
Essentially, we humans are not chickens and COVID is not the same as these MVD strains, so making such a comparison would be considered highly tenuous.
Now, maybe there could be some alien species out there that looks at New York City the same way that these researchers look at a Perdue chicken factory, but for many of us we don’t live cramped and shoulder-to-shoulder with our neighbors which may remove any trade-offs between transmissibility and virulence. MVD is also shed through the epithelial cells of chickens and can possibly linger for a period of time in the environment. SARS-COV2 spreads predominately through aerosolization and does not live long, which suggests that transmission of SARS-COV2 is predicated on living hosts and thus should not behave the same as MDV.
We’re also comprised of a diverse array of genes responsible for creating individual, distinct immunity profiles that make it difficult for viruses to find a niche within our species unlike commercial chickens which are likely to be genetically similar and thus more susceptible to immune escape.
Overall, many of the factors that likely contribute to the virulent nature of these MDV strains may not be playing out with SARS-COV2.
A leaky Leaky Vaccine Hypothesis
Like with everything, there’s quite a bit more that could be said about this chicken paper.
But let’s consider the situation we are dealing with with these current COVID vaccines.
On one hand, there are many concerns with these COVID vaccines and whether there may be long-term ramifications of conducting a global vaccination campaign in the middle of a pandemic.
On the other hand, we’re dealing with a lot of noise that makes it difficult to parse out the actual signals and the important information. The leaky vaccine study is concerning, but it may not apply with our situation with SARS-COV2. The viruses are different, and our behaviors and lifestyles are not the same as commercially-bred chickens. People who may conflate scientific studies may do so without understanding the underlying science and whether the information is applicable. We should be careful of generalizing information too far from the original source that we actually lose the context required to analyze the information. It’s hard to argue if that’s the case happening here, but considering how frequently I’ve heard of people quoting this leaky chicken study I wouldn’t be surprised if many of the people quoting it haven’t actually read the study- a problem that seems to occur quite often, and is one of the main reasons I’ve covered most of the study in this article.
At the same time the review from Bull, J. J. & Antia, R. comes with the acknowledgement that the paper was released to answer concerns about the leaky vaccine study, and so this may raise questions as to whether this paper is one that is intended to counter misinformation, or if the researchers were genuine in their criticisms of those who have latched onto the leaky chicken study and have taken it out of context.
Considering that the idea of transmissibility/virulence trade-off is one that is highly contentious, it also raises questions as to how applicable this long-used concept is in the real world or if the hypothesis is highly situational.
Keep in mind that the leaky chicken study never took into account the effects of natural immunity. If increasing virulence is a concern, we need to look at it from both the perspective of vaccine and natural infection, as well as all variations of the two- something that seems to be generally missed on both sides.
So what do I think of this study overall? I think it’s an interesting study, but I don’t think it applies too well to the situation of leaky vaccines overall. There’s quite a good deal of caveats to this study which makes it rather difficult for me to examine and extrapolate assumptions and compare them to the situation with COVID.
Bull, J. J. & Antia, R. provides a suggestion as well that is rather interesting:
The recent and ongoing Covid pandemic has revealed that re-infection is at least moderately common, whether of vaccinated individuals or individuals with a history of natural infection. It is an obvious question whether to expect evolution of a MDV-like outcome either in response to the vaccine or in response to natural immunity. For the following reasons, we suggest that it is premature to have confidence in any predictions:
SARS-CoV2 is still possibly adapting to humans as a new host; it cannot be construed as being at even a temporary evolutionary equilibrium. Furthermore, measuring intrinsic virulence is especially challenging when population immunity is changing rapidly.
Since natural immunity and vaccine immunity both wane, it will be difficult to attribute any evolution in response to waning immunity as due to a vaccine or to natural immunity.
Since most vaccines only immunize against the spike protein, evolution of escape may be easier against vaccine immunity than against natural immunity. It will be a challenge to disentangle the causes of any observed evolutionary response.
Any MDV-like outcome rests on mortality limiting transmission; the contribution of mortality to limiting SARS-CoV2 transmission so far seems to be small.
Should any high-virulence evolution of SARS-CoV2 occur, it will likely be extremely difficult to attribute separate causes to the vaccine, natural immunity and ongoing adaptation to a new host.
And so the results of leaky chicken study may be concerning, but there isn’t strong evidence that what we are experiencing with these vaccines may be similar to what is happening with commercial chickens.
Viral evolution is a stepwise process that depends on the interaction between the host, the host’s immunity, the virus’ fitness, as well as other ecological factors. As we try to rationalize what we are witnessing, remember to keep in mind that it’s the sum of its parts that dictate the direction a virus will evolve.
If you enjoyed this article or any of the other works please consider supporting Modern Discontent by becoming a paid Substack member or by donating to my Ko-fi. Your support helps independent creators publish articles outside of the mainstream. Any bit helps!

Read AF, Baigent SJ, Powers C, Kgosana LB, Blackwell L, Smith LP, et al. (2015) Imperfect Vaccination Can Enhance the Transmission of Highly Virulent Pathogens. PLoS Biol 13(7): e1002198. https://doi.org/10.1371/journal.pbio.1002198
This hypothesis may be interchanged with leaky vaccine hypothesis and may reference a model called a GMNR model. Understand that I am using them synonymously here although that may not be the case in the literature.
Bull, J. J., & Antia, R. (2022). Which 'imperfect vaccines' encourage the evolution of higher virulence?. Evolution, medicine, and public health, 10(1), 202–213. https://doi.org/10.1093/emph/eoac015
Hector, T. E., & Booksmythe, I. (2019). Digest: Little evidence exists for a virulence-transmission trade-off. Evolution; international journal of organic evolution, 73(4), 858–859. https://doi.org/10.1111/evo.13724



"We’re also comprised of a diverse array of genes responsible for creating individual, distinct immunity profiles that make it difficult for viruses to find a niche within our species unlike commercial chickens which are likely to be genetically similar and thus more susceptible to immune escape."
Well that's the critical point, isn't it? Not just leaky vaccines, but leaky vaccines during the middle of a pandemic.
All it takes now is vaccine-escape - which is already well-established with Omicorn etc - and now there is nothing stopping a highly virulent variant from circulating.
So far we've been lucky that Omicron, BA4 and BA5 have been mild, because the vaccinated don't have any real defense against them. If we happen to get a fatal variant, while 70% or so of the population only have antibodies for the long-extinct Wuhan strain, it would indeed find a massive reservoir of people with the exact same identical weakness.
Yes, I am not sure how much of this research is that applicable to SARS-CoV-2 but I am also relatively sure that nobody is trying to "weaponize" Marek's disease virus with biowarfare tech either.
Very interesting discussion!